Benötigen Sie weitere Informationen?



Benötigen Sie weitere Informationen?
The regulatory environment is becoming increasingly stringent as we strive to continually improve patient care while facing ever-increasing workloads and constant cost pressures. Ensuring quality and compliance management is now more important than ever. Working with bioMérieux on quality and compliance management gives you the confidence to know that everything is under control. Be prepared for the future .
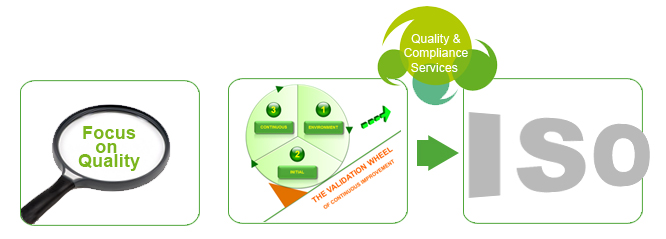
Accreditation requirements include validating your systems and methods – increasingly complex and time-consuming procedures . To help you through the “labyrinth” of validations and save valuable time, we provide you with an individualized validation package that allows you to quickly and correctly validate your bioMérieux systems step by step. We support your laboratory with:
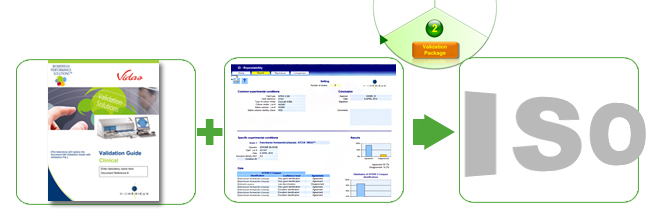
Risk analysis procedures e
We have developed guidelines tailored to your needs for implementing risk analysis in accordance with ISO 15189 regulations.
Our “Performance Solutions” offer comprehensive services – myQC solutions (Internal Quality Control (IQC)) – for immunoassays (VIDAS ® systems) that support you in complying with ISO & CAP standards. Fast and successful internal quality control (IQC) ensures the quality of the results by detecting drifts at an early stage and evaluates results by comparing data with other laboratories.
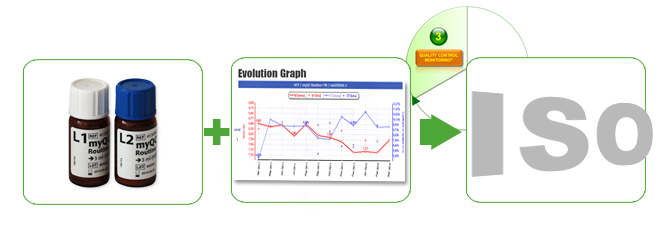
myQC for VIDAS ®
In order to enable our customers to monitor the performance data of their VIDAS systems, they have the opportunity to exchange information and experiences with other VIDAS users worldwide via the myQC platform.